Categories
Gastrointestinal disorders of cats and dogs: innovations in diagnosis and management
Gastrointestinal disorders are amongst the most common presenting complaints in small animal veterinary practice
The initial diagnostic approach to the patient with gastrointestinal should include evaluation of signalment, history and physical examination. The clinician needs to establish the time frame of the clinical signs (acute, <3 weeks, or chronic, ≥3 weeks), severity, anatomical localization (upper and/or lower gastrointestinal disease, extraintestinal disease) and identify “alarm” signs that may warrant more urgent action. This will help formulate a list of differential diagnoses and choose the most pertinent diagnostics for each patient.
The aim of this article is to compile the relevant advances in diagnosis and management of gastrointestinal disorders, including the use of biomarkers, infectious disease testing, diagnosis of low-grade intestinal lymphoma and alteration of the intestinal microbiome.
Chronic inflammatory enteropathies and biomarkers
Chronic inflammatory enteropathies (CIE) [GO(1] are characterized by chronic (> 3 weeks) or recurrent gastrointestinal signs, histologic evidence of mucosal inflammation, and the exclusion of other underlying gastrointestinal or extra gastrointestinal diseases (Washabau et al, 2010). CIE are defined by their response to empirical treatment including food responsive enteropathy, antibiotic responsive enteropathy, immunosuppressant responsive enteropathy and non-responsive enteropathy (Dandrieux, 2016). Additionally, patients with loss of protein across the gastrointestinal tract are grouped as protein-losing enteropathy (PLE) (Dandrieux, 2016). It should be noted that PLE can also be caused by primary lymphangiectasia, lymphoma or infectious diseases among others.
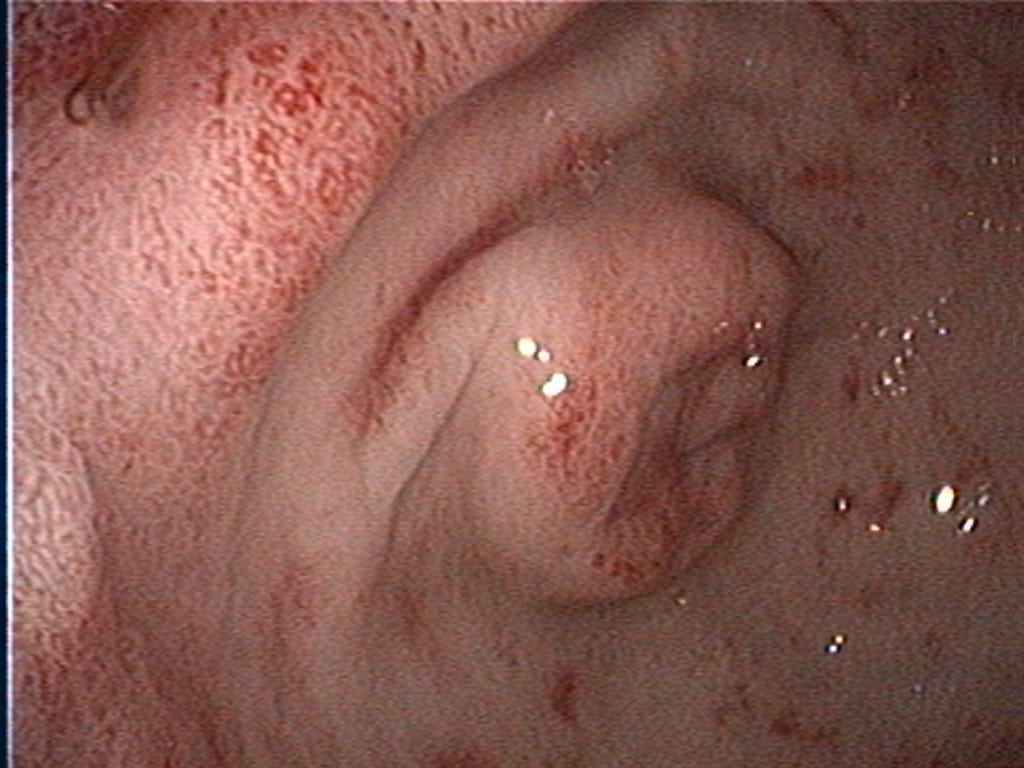
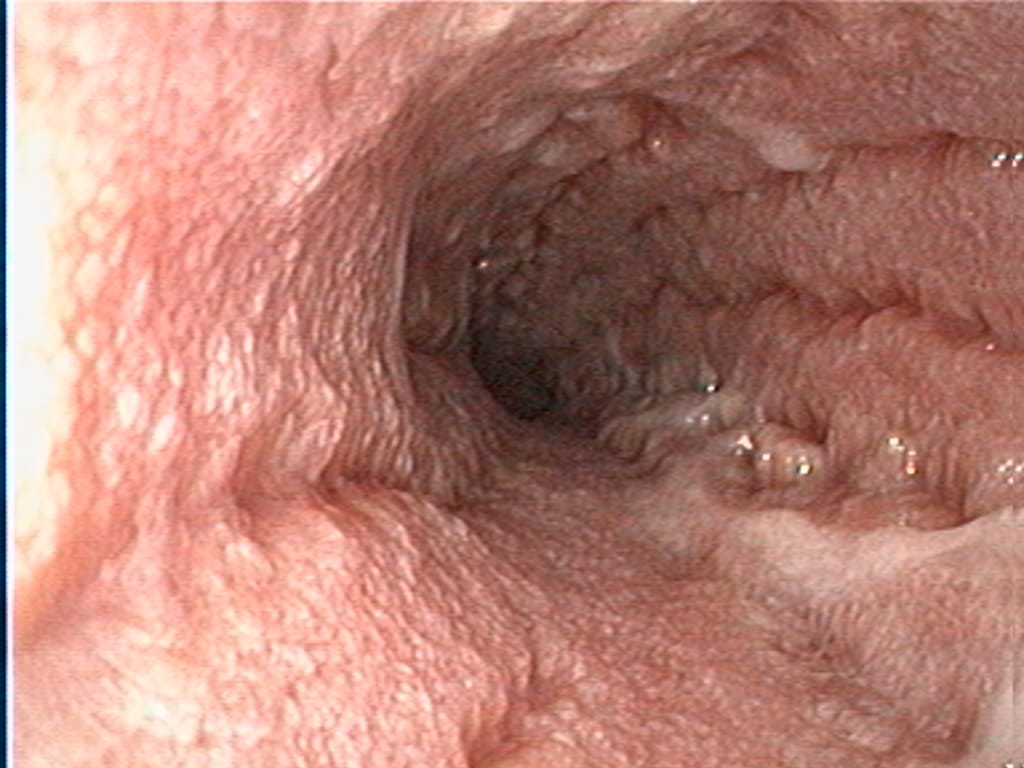
While CIE is common in small animal patients, the definitive diagnosis ultimately relies on extensive work up, including intestinal biopsies or lengthy dietary trials. Therefore, there is a special interest in developing biomarkers in patients with CIE that can aid in diagnosing, monitoring or predicting which treatment modality will benefit each patient.
KEY POINT: Several biomarkers have been evaluated in dogs with CIE. However, few are used in the routine clinical setting and further studies are needed to critically evaluate their utility
Cobalamin (B12)
It has been postulated that mucosal disease affecting the ileum reduces the epithelial expression and/or function of the B12 receptor, leading to a reduced uptake of B12 (Kather et al, 2020). Incidence of hypocobalaminaemia in dogs with CIE ranges from 19% to 38% (Berghoff et al, 2013), with a similar incidence in cats (Simpson et al, 2001). Hypocobalaminaemia has been reported as a negative prognostic factor for dogs with CIE (Berghoff et al, 2013) and these patients require parenteral or oral supplementation (Kather et al, 2020). Other causes of hypocobalaminaemia include exocrine pancreatic insufficiency (EPI) or hereditary disorders, the latter described in breeds such as Beagle, Border Collie, Giant Schnauzer and Australian Shepherd (Kather et al, 2020).
KEY POINT: A third of patients with CIE have hypocobalaminaemia and require supplementation
While the serum concentration of B12 is commonly used diagnostically, this does not directly reflect the state of B12 at the cellular level. Most reactions catalyzed by cobalamin occur in the mitochondria, and a lack of intracellular B12 leads to a reduced enzyme activity with accumulation of methylmalonic acid (MMA) and homocysteine (HCY) (Kather et al, 2020). MMA and HCY are not routinely available, although both are considered as useful markers to determine the cellular cobalamin status of dogs and cats (Kather et al, 2020).
Folate (Vitamin b9)
Hypofolataemia can result from chronic malabsorption in the proximal small intestine. However, the benefit of supplementing folate in dogs and cats with hypofolataemia has not been clearly demonstrated (TAMU, 2022). Besides, small intestinal dysbiosis may result in an increased ability of the intestinal microbiota to synthesize folate, which can lead to hyperfolataemia. It should be noted that hyperfolataemia is not predictive of a positive response to antibiotics (TAMU, 2022).
Faecal alpha 1 proteinase inhibitor (α1-PI)
Canine α1-PI has a molecular weight similar to that of albumin, and with diseases causing gastrointestinal protein loss, both are lost at a similar rate. Unlike albumin, α1-PI resists proteolysis, allowing its extraction and quantification in faecal samples (Heilmann et al, 2018 b). Increased faecal canine α1-PI concentration has been shown to reflect gastrointestinal protein loss and histologic lesions seen with PLE in dogs (Heilmann et al, 2016). To the author’s knowledge this test is currently not available in the UK.
Promising biomarkers
Biomarkers such as faecal calprotectin, calgranulin and perinuclear anti-neutrophilic cytoplasmic antibodies (pANCA) have shown promising results in differentiating sub-groups of CIE and predicting outcome. Further research is needed to evaluate the utility of these and other biomarkers in clinical practice (Heilmann et al, 2018 a).
Fluorescence in situ hybridization (FISH)
FISH can detect intact bacteria, protozoa and algae within tissues, and is readily available in the UK. This technique has been vital in the development of veterinary gastroenterology, including the identification of attaching and invasive E. coli (AIEC) causing granulomatous colitis in Boxer (Simpson et al, 2006). FISH can be requested in addition to conventional histology when the presence of enteropathogens is suspected and when there is overt neutrophilic or histiocytic inflammation. Interestingly, two studies in feline intestinal biopsies with neutrophilic inflammation identified infections with Tritrichomonas foetus and Campylobacter coli in colonic and duodenal biopsies, respectively (Yaeger et al, 2005; Maunder et al, 2016). The clinician needs to be careful when interpreting FISH results, because the presence of a positive result may represent a primary infection but also secondary overgrowth or disruption of the mucosal barrier.
Low-grade intestinal T-cell lymphoma (LGITL) in cats
Intestinal small cell lymphoma (SCL) in cats is the most frequent digestive tract neoplasia and histology consists of small, well-differentiated lymphocytes with low mitotic rates (Paulin et al, 2018). More than 90% of intestinal SCL are of T-cell origin and are currently referred as LGITL. In practice, differentiating between lymphoplasmacytic enteritis (LPE) and LGITL remains challenging. A recent study revealed some clinical and ultrasonographic variables that can guide clinicians (Freiche et al, 2021), but ultimately, intestinal biopsies are required to achieve a definitive diagnosis.
Biopsies can be obtained via endoscopy or exploratory laparotomy. Historically, the latter was considered superior to differentiate between LPE and LGITL, however, less invasive endoscopic biopsies with ancillary test can be sufficient in some cases. The main limitation to endoscopic biopsies is the localisation of LGITL, since 64% of cases have infiltration of the jejunum only, which cannot be reached endoscopically (Freiche et al, 2021 b).
Immunohistochemistry (IHC)[GO(2]
IHC involves the use of antibodies against specific antigens in tissue sections. When lymphoma is suspected, a panel of T and B-cell antibodies (eg, CD3 and CD20) can identify a monomorphic population and establish the cell lineage. Antibodies such as Ki67, pSTAT3 and pSTAT5 have been recently studied and are useful for the diagnosis of LGTIL (Freiche et al, 2021 b).
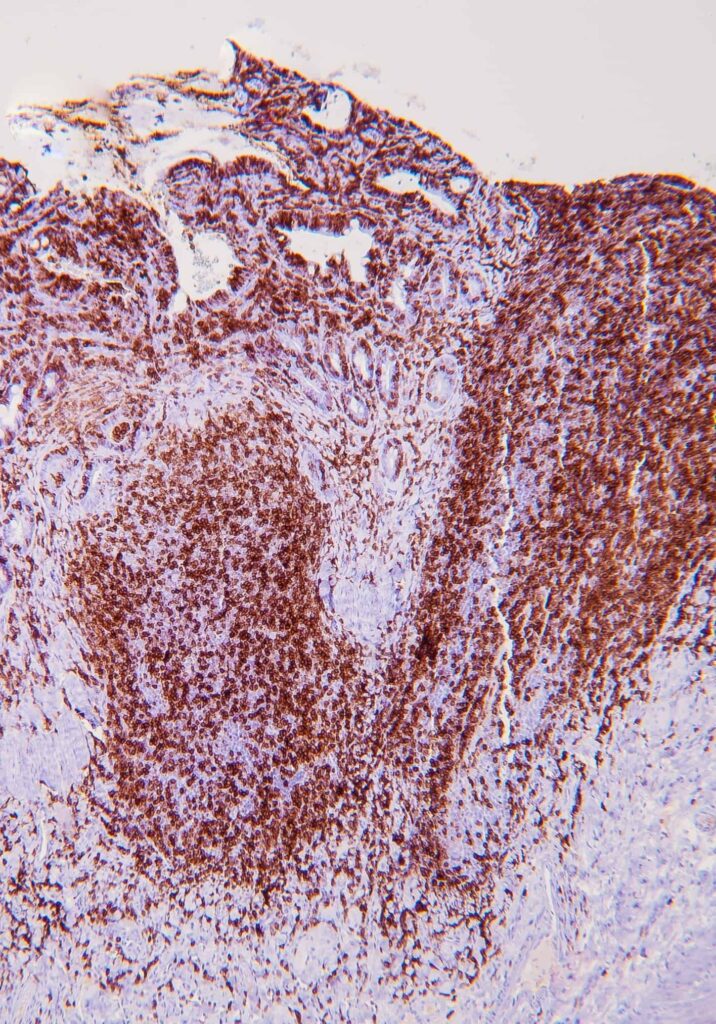
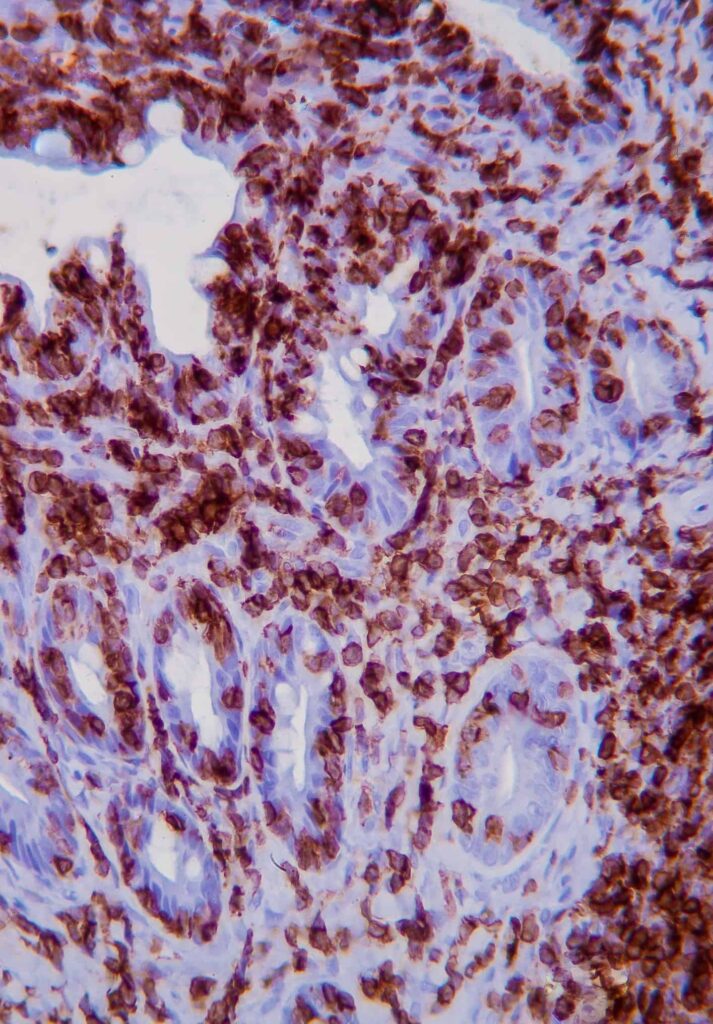
KEY POINT: The combination of histopathology and IHC is considered the gold standard for diagnosis of LGTIL
PCR for Antigen Receptor Rearrangement (PARR)
PARR is a clonality assay that uses primers sets for amplification of rearranged immunoglobin heavy chain genes and T-cell receptor gamma for B-cell and T-cell, respectively. Against what was initially thought, benign clonal expansions have been described in various inflammatory diseases including LPE in cats, where 40% of cases show monoclonality (Freiche et al, 2021b).
KEYPOINT: Clinicians should be cautious about reclassifying doubtful lymphoma based on PARR results
The intestinal microbiome
Abnormalities in gut microbial populations have been associated with a variety of gastrointestinal and systemic diseases, making the intestinal microbiome a viable diagnostic and therapeutic target. Advances in DNA sequencing and computational technology are increasing our understanding of this field but many fundamental questions remain unanswered (Barko et al, 2018).
Studies have identified decreased bacterial diversity in both CIE and acute gastroenteritis (Suchodolski et al, 2012), and have shown increased populations of potentially harmful species such Proteobacteria and Clostridium perfringens (Ziese et al, 2018). However, it remains unclear whether these changes are the cause or consequence of the disease process.
The dysbiosis index (DI) is a faecal PCR-based diagnostic test that gives a quantitative measure of the presence of bacterial groups, total bacterial numbers and summarizes them in one single number (TAMU 2022). This test is now available commercially in some UK laboratories. While there is a growing body of evidence supporting DI as a therapeutic target for cases receiving probiotics and fecal microbiota transplant (FMT) (TAMU, 2022), further research is needed to define the clinical interpretation of DI.
Probiotics
Probiotics are formulations of live organisms that confer beneficial effects on the recipient when delivered in adequate amounts. A detailed guide of the use of probiotics in dogs and cats is beyond the scope of this article and further information can be found in the literature (Barko et al, 2018; Jugan et al, 2017). We have highlighted some examples of the potential benefits of probiotics is dogs and cats with gastrointestinal disease:
- Canine parvovirus: in one small controlled study, the use of probiotic bacteria (VSL#3) reduced clinical sign, increased leucocyte counts and improved survival rates, compared with control dogs (Arslan et al, 2012). However, this study had several limitations such as the lack of faecal microbiome evaluations and the method of randomization was not described.
- Tritrichomonas foetus in cats: there is evidence to support probiotic use (Enterococcus faecium SF68 or isolated strain of Enterococcus hirae) alongside ronidazole in cats with Tritrichomonas foetus infections. A study found significant improvements in faecal scoring and body weight, as well as a reduced relapse rate (Dickson et al, 2019).
- CIE: VSL#3 showed a protective effect in dogs with CIE by decreasing clinical and histologic scores when compared with a control group that was treated with metronidazole and prednisolone (Rossi et al, 2014).
Further research is needed to fully understand the underlying mechanisms by which probiotics are effective in a range of enteropathies so that their use can be guided by an evidence-based approach.
Antibiotics
In the past, antibiotics were widely used for acute and chronic gastrointestinal disease. However, this is now debated given the age of increasing antibiotic resistance and the prolonged intestinal dysbiosis seen in healthy dogs and humans (Grønvold et al, 2010). Moreover, there is strong evidence that antibiotic use is not required in uncomplicated cases affected by enteropathogenic bacteria and acute haemorrhagic diarrhoea syndrome (AHDS) (Marks et al, 2011; Werner et al, 2020). When dealing with suspected or confirmed infectious enteropathogens, especially those with zoonotic risk (eg, Salmonella and Campylobacter), supportive treatment, use of appropriate protective equipment, and proper cleaning and disinfection are the mainstays of control (Marks et al, 2011).
KEY POINT: the majority of dogs hospitalized with AHDS and improve rapidly with symptomatic treatment only, so the use of antibiotics is not routinely recommended
It is known that a majority of patients affected by mild to moderate CIE respond to diet alone (food-responsive enteropathy), so the recommendation is to attempt dietary management in the first instance (Dandrieux, 2016). In the recent years many clinicians are moving away from using antibiotics for CIE, and these are being reserved for those patients with history of positive antibiotic response and refractory to other treatments.
Faecal microbiota transplantation (FMT)[GO(3] involves introducing faeces from a healthy donor into the gut of a diseased recipient, with the aim to modulate or replace the recipient’s intestinal microbiota (Schmitz, 2022). FMT is used in human medicine for the treatment of Clostridium difficile infections and has been shown to stabilize and increase the diversity of the microbiome (Kao et al. 2017). Early studies of this effect in veterinary medicine are promising and suggestive of a similar outcome (Sugita et al, 2019).


A single FMT administered to dogs with AHDS significantly increased the diversity of the microbiome and the population of short chain fatty acid (SCFA)-producing bacterial strains in comparison to a control group (Gal et al, 2021). However, there was no improvement in clinical outcomes post FMT and the diversity index was not significantly different 30 days post-transplant (Gal et al, 2021). In puppies with parvovirus infection, FMT did not significantly improve survival but increased the percentage of survivors who had resolution of diarrhoea within 48 hours and shortened hospitalization times (Pereira et al, 2018).
FMT is being increasingly used in refractory cases of CIE, however, the is need for prospective controlled studies to fully quantify the benefits of FMT as well as consensus regarding preparation, dosing and administration (Schmitz et al, 2022). The interested reader is referred to the current review on FMT (Chaitman et al, 2021).
Another area of growing interest is the analysis of metabolomics in dogs and cats with gastrointestinal disease. For instance, results in a study of dogs with acute diarrhoea indicated that the fecal dysbiosis was associated with significant changes in profiles of serum and urine metabolites (Guard et al, 2015). As with FMT, more research is needed to understand the importance and utility of metabolite analysis.
